BUG – Prostate Cases
CME accredited learning for treatment of prostate cancer.
This application-based learning tool (using content provided by the British Uro-oncology Group - BUG) is intended for health care professionals involved in the management and treatment of men with prostate cancer.
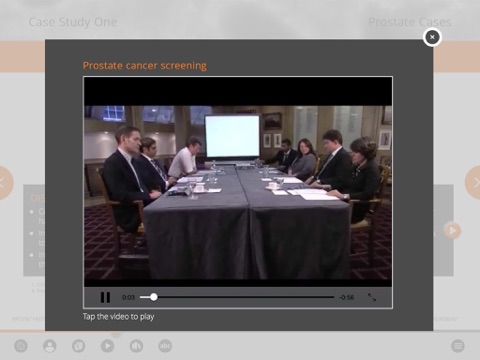
The iPad based tool contains comprehensive case studies based on real patients, but modified for the app. Each case study takes you step-by-step through the patient journey, from diagnosis through to third-line treatment and presents relevant evidence , including scans, videos of multidisciplinary team (MDT) discussions and summaries of key journal publications. At key points you will be posed with multiple-choice questions to challenge you to consider optimal management decisions.
At the end of each case study, users will be invited to offer feedback and take a knowledge test. Users who pass and who are healthcare professionals will receive a CME certificate. Each of the cases have been accredited by the European Accreditation Council for CME (EACCME) for 1 European CME Credit (ECMEC). ECMECs are widely recognised across Europe and the world, in the US, physicians may convert ECMECs to AMA PRA Category 1 Credits by contacting the AMA.
The broad learning objectives are to understand and be able to discuss:
· The key issues facing clinicians treating patients with prostate cancer.
· The management and treatment options currently available for men with prostate cancer.
· The importance of multidisciplinary teams in the management of prostate cancer.
Each case study takes approximately one hour to complete, but can be spread across more than one session. The BUG app is easy-to-use, with high quality practical content and resources presented by clinicians in an MDT setting. It is the first educational activity using an iPad app to be CME accredited by the EACCME..
The app is supported by an independent educational grant from Sanofi.